- Contact
- (919) 233-1680
- Patient Portal
-
Fertility Treatments Learn about all of our treatment options
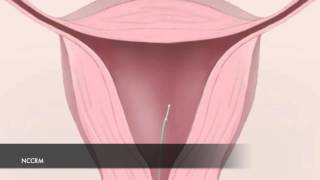
- Uterine Issues & Treatment
-
Donors & Surrogates Learn about all of our treatment options
-
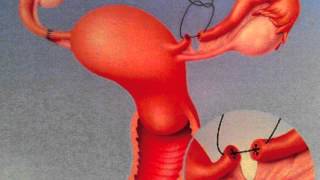
Tubal Reversal Getting Started with Tubal Reversal
-
Gynecology Services Learn about all of our gynaecology options
-
Surgical Services Learn about all of our Surgical options
-
About NCCRM Proudly serving the Triangle for over 30 years!
- New Patients
Local Patients:
For patients in the Raleigh, Cary, Durham, Chapel Hill areas, you also can call 919-233-1680 to make an appointment
at our Cary office.Out of Town Patients:
For out of town patients, please schedule a consult below or call 800-933-7202.