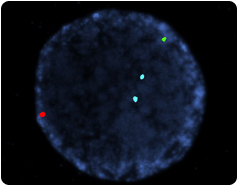
Normal Embryo
The X chromosome is labeled with a green probe, Y with a red probe, and two number 18 chromosomes with an aqua probe. This is a normal male embryo.
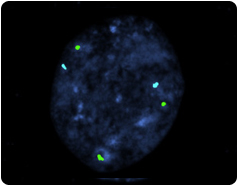
Abnormal Embryo
Three X chromosomes are labeled with a green probe; a normal female embryo would have two X chromosomes. Two number 18 chromosomes are labeled with the aqua probe.
PGD (pre-implantation genetic diagnosis) is a medical procedure in which a single cell from a 6-8 cell embryo (day 3 after retrieval) can be tested for an incorrect number of chromosomes (aneuploidy), as well as, determining the sex of the embryo. This procedure would be indicated for women with repeated pregnancy losses, for women over 37 years of age, sex selection and for couples who are known carriers of diseases that may be passed on to their children.
For women with repeated pregnancy loss and older women, the cause is often embryonic aneuploidy or an incorrect number of chromosomes in the embryo. The steps of PGD are as follows: one or two cells are removed from a six to eight cell embryo on the third day after fertilization. This is done using a microscope with very small pipits and special culture media. The cell is fixed on a microscope slide, labeled with fluorescent-linked DNA probes and examined with a fluorescent microscope (FISH). The numbers of chromosomes present are determined. At the present time, five to nine chromosome pairs that cause the overwhelming majority of aneuploidy are tested. This reduces the risk of transferring an abnormal embryo to the uterus, and increases the rate of pregnancy.
For couples with a known genetic disease, PGD is done by DNA amplification with PCR (polymerase chain reaction) instead of the FISH technique. Again the embryos are biopsied, then the cell is placed in a small test tube and PCR is used to amplify the DNA in the cell. The DNA patterns from the embryonic cell are compared with normal DNA patterns in this procedure. Over 130 different diseases can be treated with this technique.
In addition, PGD can also be utilized for family balancing or sex selection. PGD technology offers a 99.9% success rate for obtaining desired sex in a baby. This method involves In Vitro Fertilization (IVF) where the eggs are fertilized with the father’s sperm outside of the mother’s body. The resulting embryos are analyzed with PGD to determine their gender. This method allows us to determine the sex of each embryo obtained with IVF prior to returning the embryo(s) to the mother. Transferring only male or female embryos assures that the child will be of the desired gender if pregnancy results.
More Information and Techniques for Family Balancing or Sex Selection
In humans, the sex of the offspring is determined by the spermatozoa. The egg contains only X chromosomes, while a single sperm may carry either the X or the Y chromosome. If the egg is penetrated and fertilized by an X-bearing sperm then a XX female child will develop. If the egg is fertilized by a Y-bearing sperm then an XY male child will develop. This is the outcome of all normal conceptions. Although preselection of the gender of offspring has preoccupied mankind since recorded history and there have been numerous methods proposed to obtain the desired gender by certain behaviors or habits, it has only been brought near to reality since the advent of assisted reproductive technologies in the late 20th century.
In addition to pre-implantation genetic diagnosis (PGD), there is another approach to pre-determining the sex of a child by scientific techniques.
For more information on PGD or sex selection, please call (919) 233-1680 or
